-
成矿作用过程是一种复杂的动力学过程,受到地质、地球化学、地球物理等因素控制(翟裕生,1999)。地质流体作为该过程的重要载体,通过对其物理化学性质的研究有助于定量探讨地质过程的物理化学机制(段振豪,2010)。因此,热液流体中金属元素地球化学行为实验研究,是矿床学领域的关键研究内容,为研究成矿流体中物质迁移、富集及沉淀成矿机制提供重要的依据。目前,对热液流体中金属元素地球化学行为的研究方法和手段种类较多,主要包括:①基于冷封式高压釜的双贵金属管法开展热液流体中溶解度实验及热力学模拟(Qi et al., 2020;Guo et al., 2020;袁顺达等,2021;Voigt et al., 2022;Zhao et al., 2022a; 2022b);②基于金刚石压腔和石英毛细管合成流体包裹体等可视腔体与原位激光拉曼及同步辐射X射线等光谱测试方法结合的高温高压实验方法(Etschmann et al., 2010;2019;Yuan et al., 2013;2017;Zhong et al., 2015;Zhou et al., 2016;Scholten et al., 2019;Schmidt et al., 2021);③直接以钛合金反应釜为容器开展的中低温条件下的溶解度实验及热力学模拟方法(Migdisov et al., 2014a;2016;Timofeev et al., 2018;Wang et al., 2021)。其中,基于钛合金釜的溶解度实验在加拿大麦吉尔大学Williams-Jones团队得到了长期广泛的应用,为Sn-Nb-Ta-REEs-Cu-Au-Ag-Mo-Ga-W等在热液流体中的迁移、富集及沉淀成矿机制的研究提供了一系列重要的实验依据(Williams-Jones et al., 2012;2014;Migdisov et al.,2014b;Migdisov et al., 2016)。近年来,中国高度重视实验矿床学的研究,国内多个大学及研究院所与Williams-Jones团队开展了广泛的合作研究,并在国内多个实验室建立了这套技术方法,。该技术方法并通过热液流体中W、Mo及Sc等金属的溶解度实验及热力学模拟,识别出一系列重要金属络合物形式及其热力学参数,并通过成矿过程的模拟,在矿床成矿机制的研究方面取得了一系列重要认识(Wang et al., 2019;Shang et al., 2020;Wang et al., 2022)。
近年来,战略性关键金属矿产的成矿作用研究和找矿勘查已成为国际矿床学关注的热点(Hu et al., 2012;Mao et al., 2019;2021;Zhao et al., 2022c; 2022d)。其中,各种关键金属超常富集机制是当前备受关注的关键科学难题(Yuan et al., 2018;陈骏,2019;2019;温汉捷等,2019;2020;毛景文等,2019;袁顺达等,2020;熊小林等,2022;蒋少涌等,2022)。鉴于钛合金釜的溶解度实验和模拟方法在热液流体中金属元素地球化学行为研究中的突出优势,有望成为解决该科学难题的重要手段。因此,本文在全面介绍各类热液实验方法的优点及应用局限的基础上,着重介绍钛合金反应釜溶解度实验方法及热力学参数计算过程,尤其详细地介绍了HCh模拟软件的应用,并结合已有的钪溶解度实验成果在成矿过程研究中的初步实践,全面系统地展示该方法在成矿过程研究中的重要潜力,以期推进该方法广泛应用于热液矿床中各类战略性关键矿产元素超常富集机制研究。
1溶解度实验研究现状长期以来,溶解度实验研究极大程度促进了地球化学的快速发展。尤其进入21世纪以来,热液模拟实验研究在装置和技术方面已取得突破性进展。采用钛合金反应釜开展的溶解度实验研究,在络合物结构及热力学参数的计算方面发挥着重要作用,积累了大量研究成果,例如Migdisov等(2014b)在200~400℃、1000 bar条件下研究了稀土元素在含氟流体的迁移沉淀机制;Migdisov等(2014a)在350~550℃条件下对低密度、含HCl水蒸气相中铜的迁移进行了预测;Timofeev等(2017)在100~250℃进行实验测定了钽在含氟流体的溶解度;Nisbet等(2018)在150~250℃条件下研究了含氯流体中钍的溶解度。最近Williams-Jones团队通过实验和软件模拟探讨了蒸汽流体在斑岩Cu-Au-(Mo)系统成矿过程中的,位素分馏展开研究,刻画了银从液相到气相的反应模型,也测定含氟溶液中钪的溶解度和络合物种形(Wang et al., 2022)。此外,依托原位观测技术,Pokrovski团队结合XAFS分析(X射线吸收精细结构谱)开展钛高压釜溶解度实验,通过X射线衍射和电子显微镜检测反应器壁上形成的致密晶团与硬壳,获取结晶度和晶体形态等信息,用电感耦合等离子体发射光谱(ICP-OES)分析XAFS运行前后溶液中金属浓度及溶解情况,而后基于密度模型和HKF状态方程对溶解度数据进行分析(Pokrovski et al., 2005a)。Pokrovski团队采用该方法研究了热液流体中Sb-Ge-Au-Ag等金属的赋存形态,对水-盐-气体系H2O-NaCl-KCl-HCl在水热条件下的金属气液分配,硫配体对金属气液分配的影响(350~500℃条件下H2O-S-NaCl-KCl-NaOH体系)展开研究讨论(Pokrovski et al., 2000;2005a;2005b; 2006;2008;2009;2013);近年来Pokrovski等(2015)也利用原位拉曼光谱测定了热液中S
 的稳定性与丰度。Zajacz团队则是通过在原位开裂石英片中捕获流体相的合成流体包裹体(SFI)方法进行采样,其中含有实验相组合的胶囊由贵金属合金管制成,实验时利用激光烧蚀电感耦合等离子体质谱(LA-ICPMS)分析获取金属的溶解度与流体/熔体分配系数等数据,实验的反应容器种类多样,包括Nimonic 105合金冷封压力釜、钛-锆-钼(TZM)反应釜以及活塞圆筒实验装置等(Zajacz et
al., 2008;2010;2013;2015;Yin et al., 2018)。在此条件下,对Au、Cu、Ag的溶解度,共存流体和硅酸盐熔体中的元素分配,Cu-Sn-Co-Ag气相输运与沉积(Hunter et al.,
2020)开展了广泛研究。基于动力学模拟(MD)方法和X射线吸收光谱,Brugger团队研究了Cu-Au-Te-Bi-Zn-Eu络合形态和热力学性质(Brugger et al., 2007; Mei et al., 2014; Zhong et al., 2015;
Etschmann et al., 2016; Mei et al., 2016; Liu et al., 2017),最近也对硫在热液中钼迁移中的作用以及富氯热液中钇的络合开展了相关研究(Liu et al., 2020; Guan et al., 2020)。
的稳定性与丰度。Zajacz团队则是通过在原位开裂石英片中捕获流体相的合成流体包裹体(SFI)方法进行采样,其中含有实验相组合的胶囊由贵金属合金管制成,实验时利用激光烧蚀电感耦合等离子体质谱(LA-ICPMS)分析获取金属的溶解度与流体/熔体分配系数等数据,实验的反应容器种类多样,包括Nimonic 105合金冷封压力釜、钛-锆-钼(TZM)反应釜以及活塞圆筒实验装置等(Zajacz et
al., 2008;2010;2013;2015;Yin et al., 2018)。在此条件下,对Au、Cu、Ag的溶解度,共存流体和硅酸盐熔体中的元素分配,Cu-Sn-Co-Ag气相输运与沉积(Hunter et al.,
2020)开展了广泛研究。基于动力学模拟(MD)方法和X射线吸收光谱,Brugger团队研究了Cu-Au-Te-Bi-Zn-Eu络合形态和热力学性质(Brugger et al., 2007; Mei et al., 2014; Zhong et al., 2015;
Etschmann et al., 2016; Mei et al., 2016; Liu et al., 2017),最近也对硫在热液中钼迁移中的作用以及富氯热液中钇的络合开展了相关研究(Liu et al., 2020; Guan et al., 2020)。
综上,多元的高温高压技术以及分析测试方法为深入认识流体中金属迁移富集机制及定量化模拟成矿过程创造了条件。以钛合金反应釜为容器的实验方法是传统的金属元素溶解度研究方法,在国内外多个课题组中得以应用。
2热液流体成矿实验流体是热液矿床成矿过程物质迁移的重要介质之一,是研究成矿过程的精髓。目前,针对元素在热液流体中迁移形式的研究主要包括离位分析与原位分析(赵晓莉,2013)。前者以反应釜为容器,结合激光剥蚀电感耦合等离子体质谱仪(LA-ICP-MS)用于固体及液体样品微量元素分析,后者利用可视腔体结合原位的紫外-可见(UV-vis)光谱、激光拉曼光谱、核磁共振(NMR)光谱、中子衍射和散射方法、X射线衍射和X射线吸收(XAS)光谱等分析技术(Grunwaldt et al., 2003),直接测定络合物的结构及相关物化参数。不同实验方法、分析方法均具有各自的优点和局限性(见表1、表2),需要针对具体的科学问题,选择合适的方法。
2.1实验方法实验矿床学研究方法颇多,针对岩浆-热液成矿过程的实验模拟通常采用以下几种方法:冷封式高压釜的双贵金属套管法、钛合金反应釜溶解度实验方法、可视腔体原位观测法(表1)。其中冷封式高压釜实验方法基于矿物中捕获的流体包裹体(FIs)信息,可以模拟合成流体包裹体(SFIs)与热液流体反应,研究流体中的金属元素迁移(Qi et al., 2020)。并且基于该方法制备实验样品复现天然样品,可还原地质条件,在地质灾害监测上有示警作用(Voigt et al., 2022)。此外,通过该方法研究熔体-流体相间分配系数,可指示金属成矿(Zhao et al., 2022a、b)。虽然该方法能够满足高温高压条件的实验研究,但焊封的贵金属管容器容量相对较小,且淬火后很难快速将固体与溶液分离,淬火过程中沉淀于贵金属管壁的金属难以有效回收。其次是以金刚石压腔、熔融石英毛细管胶囊(FSCC)以及石墨反应腔等“可视腔体”为反应容器的,此类实验装置的突出优势是与原位观测结合,能够直接获得高温高压条件下各类金属元素在流体或熔体中络合物结构信息,其中利用HDAC开展熔体包裹体均一实验得到广泛应用(李建康等,2011)。近年来,为攻克低温条件下氧化还原反应控制的难题,研究者将样品密封在FSCC中,并放入HPOC(高压可视反应腔)中,待反应装置升温至目标温度,开启氧逸度控制装置通入氢气实现氧逸度控制,随后结合拉曼光谱技术实时获取原位分析数据(Chou et al., 2021b)。基于钛合金釜直接作为反应容器的溶解度实验方法,对于不同流体体系中金属元素迁移机制的研究有重要意义,成果丰硕,在下一节中将展开系统介绍。
表1 研究金属络合物在热液流体中地球化学行为最常用的实验方法(据Chou,2021a修改)
Table 1 The most commonly used experimental methods for studying the geochemical behavior of metal complexes in hydrothermal fluids(modified from Chou,2021a)
实验方法
反应装置
主要优势
主要局限性
钛合金反应釜溶解度实验
钛合金反
应釜
明确决定反应过程的关键参数,精确测定金属/配体摩尔比值;元素溶解度可结合ICP-MS测定检测限低;反应容器体积大,检测参数更全面;淬火过程使固体与流体快速分离,沉淀物处理简便
需要合适的矿物质;要求精确控制许多实验条件与实验参数;没有关于络合物几何信息;溶液取样/淬火造成的人为干扰
冷封式高压釜的双贵金属套管法
冷封式高压釜与合金
胶囊
可以模拟高温高压的成矿过程(温度高达850℃,压力20 MPa),可利用LA-ICP-MS、ICP-MS测定溶解度
淬火后才能观测实验产物;以双贵金属管为容器,溶液体积小,实验后的流体成分较难回收;低温条件(400℃以下)下贵金属反应管扩散十分缓慢无法控制氧逸度
可视腔体原位观测法
热液金刚石压腔
可提供更高温度和压力的实验条件;能进行原位观测分析手段多样;装置体积小,便于操作;能够控制冷却速率
样品体积小;不能直接测量压力,且对样品压力不能自由变化;不易加载气体样品;金刚石信号和荧光的干扰会使金刚石收集的拉曼信号减弱
表2 研究金属络合物结构及组成的常用分析方法(据Brugger et al., 2016修改)
Table 2 Common analytical methods for studying the structure and composition of metal complexes(modified from Brugger et al., 2016)
分析方法
主要优势
主要局限性
数量
电化学
原位,研究氧化还原过程有利
电极表面可能发生化学变化
少量、
定量
紫外可见光谱(UV-vis)
原位,仪器简单对配位过程中的电子跃迁十分敏感
偏离Beer-Lambert定律会影响定量解释,不是所有的络合物都有有效的光谱特征(饱和的碳氢化合物,如甲烷、乙烷等)
少量、
定量
振动光谱(IR-NIR &Raman)
原位,对电子敏感。原位光谱;Raman适用于高压(激发体积小,适用于金刚石腔研究)
Raman:要求高浓度溶质;对不同络合物应用不全面(键能对应的波数在1~100 cm以上)
少量、
定量
核磁共振(NMR)
原位,对配体交换动力学高度敏感
由于样品需要放置在超导磁体中要求低温条件,对大部分
核敏感
少量、半定量
中子/X射线衍射和散射
原位,基于中子方法对氢高度敏感
需要集多方法配合;原子序数较高元素对X射线衍射更敏感
—
XANES & EXAFS
原位,光谱可从第一性原理精确地计算关于配位几何的直接信息
有些元素由于边缘能量较低(如Cr、Ti)而难以获得,有些配体很难区分(例如O/F,Cl/S)
少量、(半)定量
ICP-MS & LA-ICP-MS
检出限低;元素覆盖范围宽,具备测量同位素变化的能力;适用有机溶剂;样品需求量少;可快速扫描分析
运行费用相对较高;样品介质的影响大;剥蚀过程中的元素分馏效应;存在质谱干扰
大量、高质量
2.2分析方法随着实验技术和实验设备的发展,溶解度数据的测定方法更加多样,主要分为原位观测和离位分析两种方法。下面分别进行简要介绍。
原位分析方法多样,包括:电化学、紫外可见光谱、振动光谱、核磁共振、中子/X射线衍射和散射和XANES & EXAFS(表2)。其中,应用最早的电化学方法常用于研究氧化物的电子结构以及氧化还原过程(Lin et al., 2014)。然而,电化学分析时需要将物质本身当作一个特殊电极,在测量过程中电极表面电化学信息会被掩盖,限制热力学数据的测定(Lange et al., 1993)。相对于电化学方法,紫外可见-吸收光谱反映价电子跃迁,常用于过渡金属配位化学的研究,若电子从配体到金属的跃迁非常强烈,对络合物浓度的检出限可低至10-6级别,而当d-d跃迁能量低时,光谱将无法识别出络合物结构(Suleimenov et al., 2000; Brugger et al., 2016)。因此,紫外光谱对于特定电子跃迁研究存在一定的局限性。振动光谱主要有2种:拉曼光谱和红外光谱,可以确定复杂的晶体结构,是水溶液中络合物研究的常用分析手段,在相态识别等方面有突出优势(Gillet, 1996)。并且,基于密度泛函理论(DFT)对数据进一步处理,通过电子结构计算,可以得到矿物的热力学稳定性和振动光谱(Efthimiopoulos et al., 2017),进而实现理论建模。然而,拉曼分析需要络合物本身具有强拉曼活性及较高的浓度,当实验温度过高时荧光效应有碍光谱分析(Reynard et al., 1994)。
核磁共振则是另外一种强有力的原位分析物质结构的方法,可以探测配体交换与络合物形成动力学参数,但由于磁铁高温退磁限制实验温度(≤150℃)(Cusanelli et al., 1996; Grundler et al., 2013)。中子衍射和散射方法因中子极强的穿透力适用于极端条件下的原位观测,并且,对磁性物质研究有明显优势(Xia et al., 2017)。此外,中子衍射还能区分热运动信息与结构细化中的位置占用信息(Rinaldi, 2002; Dove, 2002; Rinaldi et al., 2005),但是中子衍射波相对较长,导致其分析结果偏离真实值且较为分散。基于这些限制因素,上述两类分析方法在热液成矿研究的应用较少。
X射线是一种非常重要的原位物质结构分析技术。其中,X射线衍射不仅可以获得晶体内部原子分配规律实现晶体结构分析(Waychunas et al., 1990),而且还可提供溶液和溶质径向分布函数信息(Neilson et al., 2001)。X射线吸收(XAS)光谱的可探测到核能级电子的激发,适用于水溶液配位化学研究(Bunker, 2010)。但是,在高压高温条件下探测器收集大散射角有限制,需要高浓度溶质,并且热液流体的高温与侵蚀性会限制XAS测量的X射线光谱。根据吸收边的位置,将X射线吸收精细结构分为X射线吸收近边结构谱(XANES)和扩展X射线吸收精细结构谱(EXAFS)。其中,XANES可以补充EXAFS的数据(Benfatto et al., 2002; Testemale et al., 2004),对原子的电荷分布与配体信息敏感,广泛用于高温原位化学实验以及金属络合物稳定常数的测定(Liu et al., 2007; 2011)。而EXAFS可反应中心原子周围的径向电子分布特征。但X射线只适用于部分原子,对配体的鉴别也存在一定局限性。
ICP-MS(电感耦合等离子体质谱)和LA-ICP-MS(激光剥蚀电感耦合等离子体质谱法)是一种无机元素分析技术,能分别对淬火后的溶液、固体产物精准测定,且动态范围很宽(>10个数量级),具有极低的检出限。其中LA-ICP-MS直接用于固体样品分析,激光剥蚀损害小属微损分析。
3钛合金反应釜溶解度实验方法钛合金反应釜溶解度是通过获取金属浓度与T、mL (L代表F-,Cl-等阴离子)、pH、
 (H2O)、
(H2O)、 (HCl)、
(HCl)、 (O2)等,在不同的热液流体系统中达到反应平衡后各组分含量(郑海飞等,1983),结合观测数据来模拟成矿的迁移富集机制,为成矿作用研究提供理论支撑与实验依据(刘吉芳等,1984;何江,1993;张生等,1995)。
(O2)等,在不同的热液流体系统中达到反应平衡后各组分含量(郑海飞等,1983),结合观测数据来模拟成矿的迁移富集机制,为成矿作用研究提供理论支撑与实验依据(刘吉芳等,1984;何江,1993;张生等,1995)。
钛合金反应釜法直接将钛合金釜作为反应容器,可在釜体内部同时放置固体反应物和氧化还原缓冲剂,不仅可以有效控制中低温条件下体系的氧逸度,还可以实现实验流体样品的高效回收。结合等离子体质谱技术(ICP-MS)和发射光谱分析(ICP-OES),可以测定回收流体的微量元素组成,同时利用质谱法也可以分析同位素组成(Liebscher, 2007)。基于ICP-MS技术获得的元素配位比范围更广,检测限可以达到0.1×10-9,有利于构建成矿模型(熊小林等,2022)。因而,这种方法在对热液流体中溶解度极低的元素的溶解度及络合行为的研究具有独特优势。并且,通过设定浓度梯度,改变初始流体组成,使得热力学模拟配体与金属络合的信息更全面。与利用原位光谱或波谱直接获取络合物结构信息不同,基于这种实验方法得到的大量溶解度数据,可以用来有效推导在热液体系中元素迁移沉淀的热力学模型(Brugger et al., 2016)。得益于钛合金反应釜溶解度实验的多组分、多梯度、多变量等特点,其在定量模拟成矿过程中数据分析更全面。
3.1实验原理溶解度实验是一种测定固体物质在溶剂中溶解度的实验方法。通过一系列实验推测金属络合物,计算相关热力学参数可以进一步模拟成矿过程,其中热力学第二定律是计算热力学参数和模拟实验的理论基础:
$${\mathrm{d} G=V \mathrm{~d} P-S \mathrm{~d} T+\sum_{i=1}^{K} \mu_{i} \mathrm{~d} N_{i}}$$式中,G代表吉布斯自由能,V为摩尔体积,P表示体系压力,S为熵,T为热力学温度,μi为偏摩尔吉布斯能,Ni为体系中的粒子数,K为体统中组分数量(Cottis et al., 2010; White, 2013)。
在热液系统中,金属元素主要以各种络合物形式存在于热液流体,而络合物在流体中的配体和配位数决定溶解度,为更好地利用实验获取的溶解度来解译元素的迁移行为,需要结合配位化学的理论基础来理解热液流体中金属元素的地球化学行为。配位化学是研究金属原子或离子与有机、无机的离子或分子反应形成配位化合物的科学。在溶解度实验中通过测定反应平衡后溶液中金属元素溶解的浓度,确定该金属离子以配位键结合配体的数目(Han et al., 2023),以钪的羟基络合物为例:
[Sc]=[Sc3+]+[Sc(OH)2+]+[Sc(OH)2+]+[Sc(OH)30]
由此,Sc与OH-结合形成络合物的配位数(n∑[Sc(OH)n3-n])可通过以下公式换算得出:
$$\vec{n}=\frac{n \sum\left[\mathrm{Sc}(\mathrm{OH})_{n}^{3-n}\right]}{[\mathrm{Sc}]}$$此外,Pearson于1963年提出的软硬酸碱理论(HSAB),也是推断络合物中金属离子与配体组合稳定性的重要理论支撑,其原理为硬(不可极化)阳离子将优先与硬阴离子(离子键)结合,软(可极化)阳离子将优先与软阴离子(共价键)结合。目前,热力学第二定律和配位化学仍是认识金属络合行为的重要理论基础,Pearson软硬酸碱理论也是理解和预测热液流体中金属元素迁移行为的理论依据。
3.2实验方法流程基于上述对比分析,钛合金釜溶解度实验方法对于研究金属元素在热液流体中的迁移、富集及沉淀行为具有独特的优势。为了促进该方法在热液金属矿床成矿作用研究中的应用,我们下面将系统介绍实验及热力学模拟的方法流程。
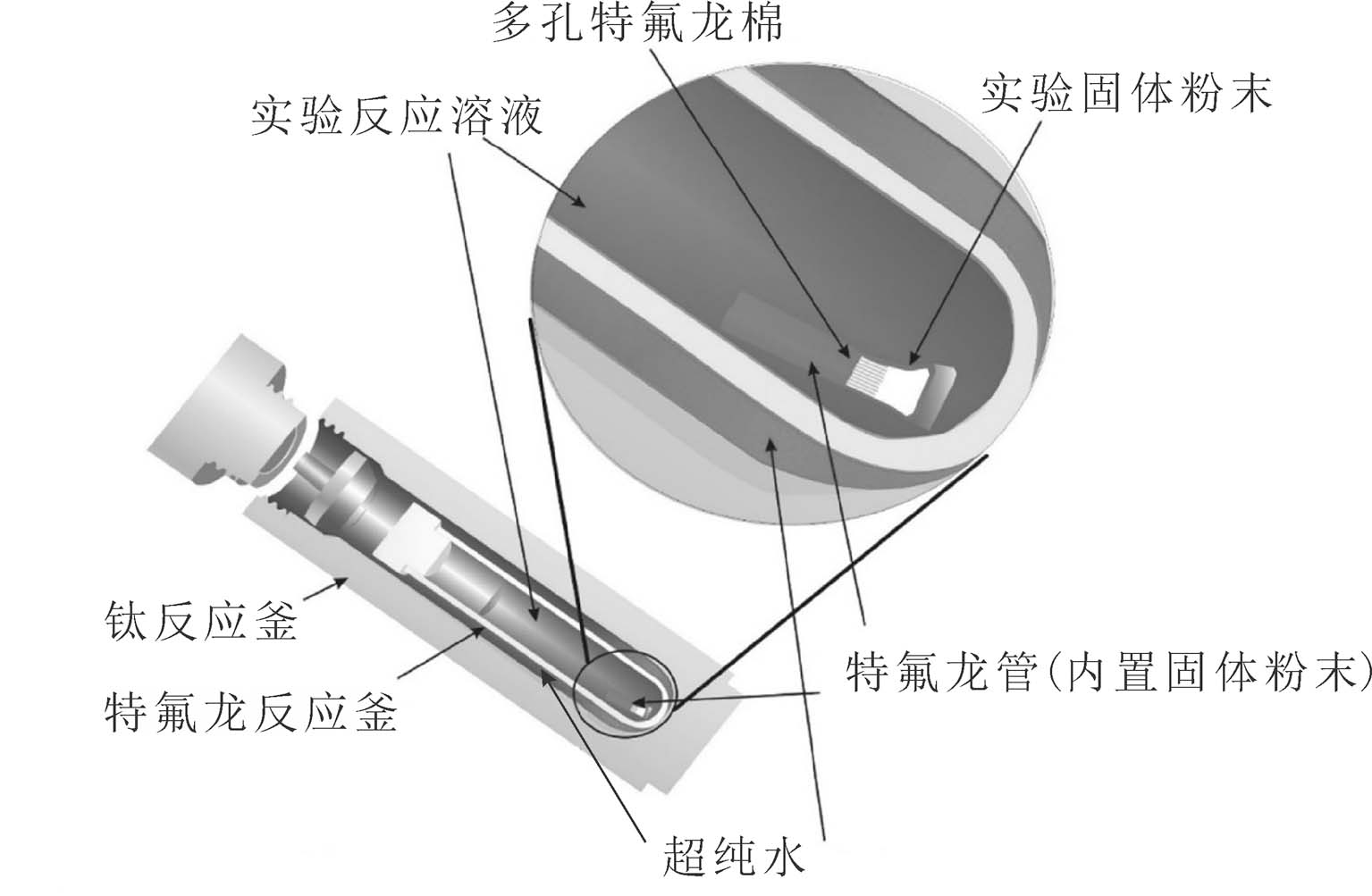
实验所用设备包括高温加热炉、钛合金反应釜(图1)、特氟龙反应釜(含氟体系时使用)和K型热电偶等。
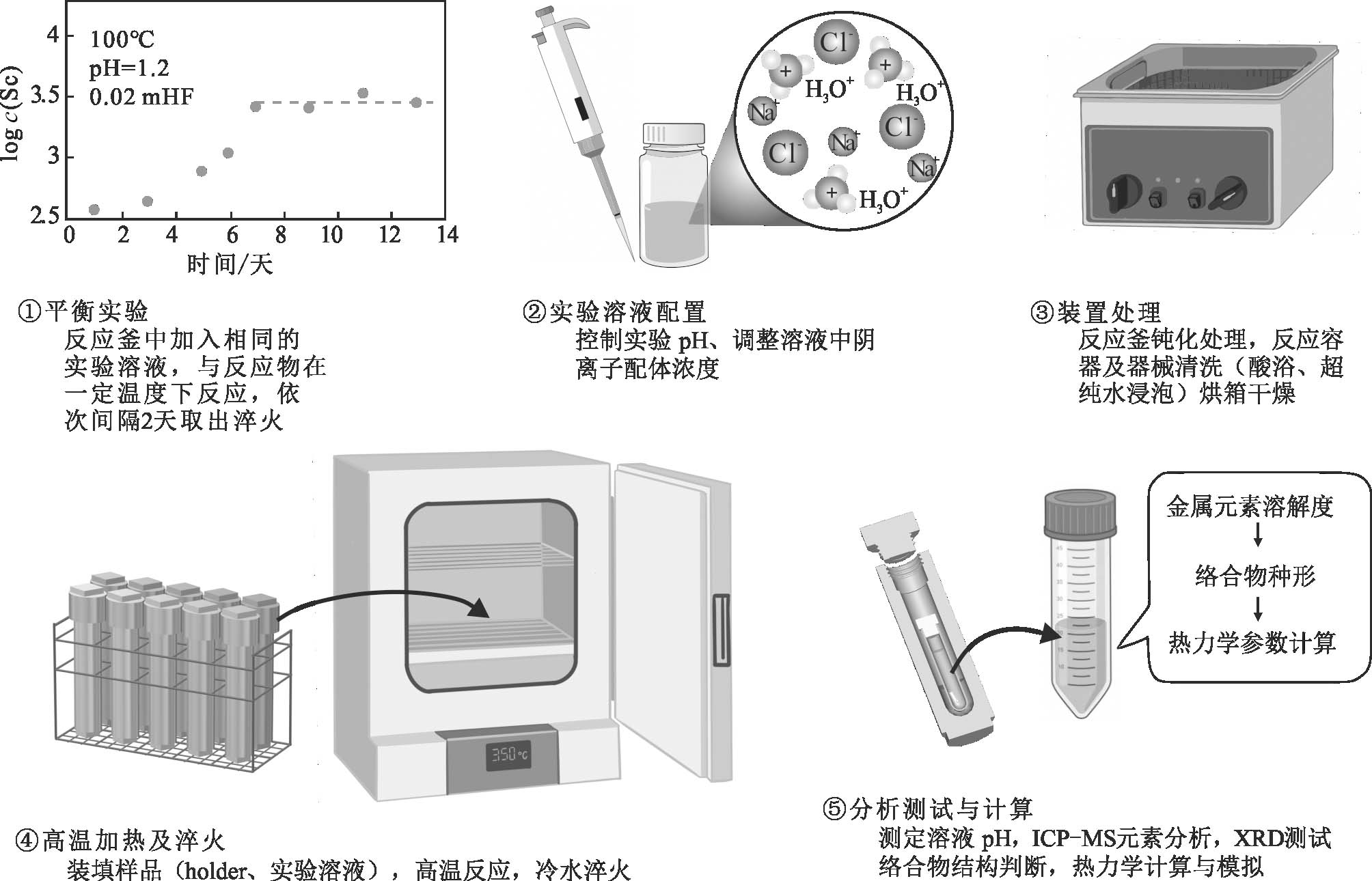
实验前期的准备工作主要包括反应容器预处理和平衡实验。首先对耐腐蚀高温高压钛合金反应釜进行钝化处理,并将钛合金反应釜等与反应溶液直接接触的装置进行24 h酸浴,再用去离子水浸泡24 h后进行干燥处理。其次,实验开展前为确定体系达到平衡的时间需要提前进行平衡实验,最低实验天数为目标元素浓度稳定(在误差范围内波动)所需时间。在实验中保持反应釜中实验溶液浓度、pH以及反应炉中反应温度不变,仅改变反应时间。实验持续时间从1天到14天不等,分别间隔2天取出反应釜淬火并对反应后的pH值和溶质浓度进行测定,由此可获得待测金属元素浓度与时间的变化曲线,溶液中元素浓度达到最大并趋于稳定的天数即为实验达到平衡所需的时间。
实验开始先向“holder”(由石英管或特氟龙管制成)中加入反应物固体粉末,利用移液枪沿反应釜内壁加入实验溶液,利用氩气排出反应釜中空气后密闭再放入预热至实验温度的反应炉中。经过一定反应周期后体系到达平衡状态(持续时间由平衡实验确定),将反应釜快速取出并放置在冷水中进行淬火。待反应降至室温后可处理实验溶液,取3 mL反应釜中的溶液测定pH和阴离子配体的含量,随后沿反应釜内壁逐滴加入定量的硝酸(或其他浓酸)清洗沉淀物,静置至少2 h后取出溶液用于测定金属元素浓度。当实验体系为含F体系时可在钛合金反应釜中加入特氟龙内衬,将实验的样品与溶液放置在特氟龙管中反应,并且,封闭钛合金反应釜前,需要在钛合金反应釜与特氟龙反应釜之间加入适量去离子水以保证加热及淬火过程中内外压的平衡(实验步骤参见图2)。
图1 实验反应装置示意图(据Wang et al., 2022修改)
Fig. 1 A sketch of the experimental set-up (modified from Wang et al., 2022)
3.3热力学参数计算方法在溶解度实验中热力学常数计算通常包括:络合物与离子活度的计算、络合物平衡常数和形成常数的计算。
(1)络合物与离子活度的计算基于1923年提出的Debye-Hückel离子互吸理论,在溶解度实验反应过程中,配备的起始溶液与成矿金属元素反应获得反应平衡溶液,当溶液模型处于动态平衡时,利用离子强度计算溶液中络合物及离子的活度,计算方程为(Helgeson, 1969;1981):
$${\log \gamma_{n}=-\frac{\mathrm{A} \cdot[z_{n}]^{2} \cdot \sqrt{I}}{1+\mathrm{B} \cdot \mathfrak{a} \cdot \sqrt{I}}+\mathrm{b}_{\gamma} \cdot I}$$式中,Ⅰ代表实验溶液整体离子强度,z为实验溶液中络合物或离子的价态,A和B参数为方程常数,å为实验溶液中离子间最小间距,b为溶液扩展参数。在不同的流体体系实验中,实验溶液中络合物的形式也存在差异。因此针对不同的阴离子配体及其络合物种类展开研究,可以通过定量溶液中阴离子浓度来讨论不同络合物形式的溶解度变化。
为了获得有效的热力学形成常数和溶解度常数,根据Nelder-Mead simplex模型(Nelder et al., 1965; Dennis et al., 1987)对最小化误差函数进行计算,其表达式为:
图2 钛合金反应釜溶解度实验流程图
Fig. 2 Flow chart of solubility experiment in titanium alloy reactor
$$U=\sqrt{\sum_{i}\left(\frac{\log C_{M}^{\text {Theo }}-\log C_{M}^{\text {exp }}}{\log C_{M}^{\exp }}\right)^{2}}$$式中表示数据库中的第个实验点,是实验测定的金属元素(根据实验研究对象替换)的浓度,是用理论计算出来的浓度(Migdisov et al., 2007; Timofeev et al., 2015)。
络合物形成常数计算,需要结合金属阳离子、阴离子已有的吉布斯自由能及实验获得的两者浓度关系,通过如下状态方程获得:
ΔGr(T) =ΔGproduct(T)ΔGreactant(T)
ΔGr(T) =-RTlnβ
式中ΔGr(T)为反应方程吉布斯自由能,R为常数,T是实验温度,lnβ是形成常数。
此外还可通过HCh软件的“Unitherm database”功能依据反应方程得到Helgeson-Kirkham-Flowers (HKF)模型状态方程或Ryzhenko-Bryzgalin(RBM)模型状态方程,进一步得到络合物平衡常数logK。
3.4模拟程序地球化学系统的物理化学过程模拟需要了解体系组分的热力学性质,通过程序可以高效地计算推演反应平衡状态模拟预设的地球化学系统,计算机算法具有极高的准确性(Oelkers et al., 2009)。从实验数据中获得流体组分的热力学性质,并通过程序代码进行热力学计算是地球化学建模的关键。表3列举了几种模拟程序,通过结合热力学数据库可快速计算出各种地球化学体系中的矿物溶解度和溶质络合物形式。
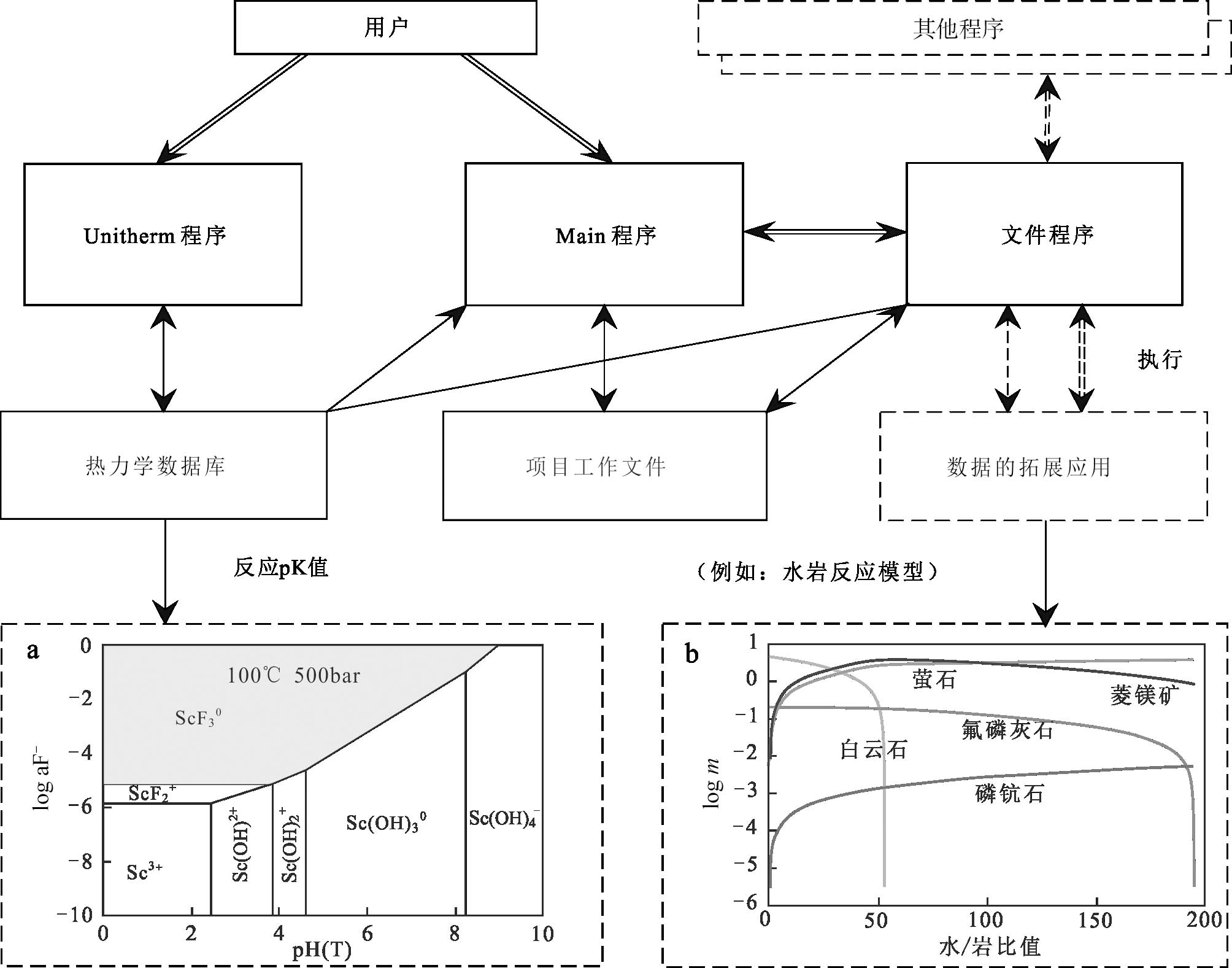
成矿过程模拟可以定量化描述金属元素在流体中的迁移富集及沉淀过程,本文主要基于HCh软件介绍成矿模拟过程,HCh可以模拟不同的体系中多组分的化学平衡,是溶解度实验中常用的研究方法之一。目前在程序中主要应用的两个功能为数据库程序(Unitherm database)和主程序(Main program)(图3)。
数据库程序(Unitherm database)设计主要用于编制和处理热力学数据库,添加反应物(Items)以及反应方程(Reaction)都是通过该功能实现,而地球化学系统和过程的模拟是通过主程序(Main program)创建模拟文件进行的,主要内容包括:系统组成(System)、系统反应物(Blank)、参与反应的溶质与溶液(Input)、模拟的控制流程(Control)等,其中以“Control”的应用较为突出,常用于模拟算法和处理模拟结果。应用时利用“Items”添加反应物,“Reaction”输入反应方程,来获取不同温压条件下反应的pK值(图3)。也可通过设置“System”和“Blank”文件来设置体系中组成成分,随后计算时启动吉布斯程序(Gibbs program)模拟即可,它会利用主程序编制的文件与数据库(Unitherm)中的络合物性质来计算离子活度,进而完成多种热力学计算,其中反应的离子强度会受到体系内的主导组分和pH控制由系统内置影响因子(b-gamma)决定,反应进程则由“Control”文件预设的地质过程控制。
基于实验测定数据与计算的络合物热力学参数,可以探讨不同温度和pH条件下热液流体中络合物相对含量的变化,也可以结合已有的络合物热力学数据绘制相图(如图3a),由此可直接获取不同条件下(温度、压力、pH等)热液流体中主导金属元素溶解度的络合物种形。在流体-岩石相互作用过程中,金属元素浓度也不断发生改变,结合络合物的溶解度可以模拟水岩反应中金属元素的沉淀过程(如图3b)。当前模拟软件的开发愈发成熟,热力学数据也在不断更新完善,未来这些计算程序将在更好地了解金属的能量性质及其在热液中的行为方面发挥越来越重要的作用。
表3 地球化学模拟程序(据Oelkers et al.,2009修改)
Table 3 Geochemical modeling codes (modified from Oelkers et al.,2009)
程序
下载地址
SUPCRT92
http://pdukonline.co.uk/download
+SLOP98 database
http://geopig.asu.edu/supcrt_data.html
EQ3/6
https://ipo.llnl.gov/technology/software/softwaretitles/eq36.php
PHREEQC
http://wwwbrr.cr.usgs.gov/projects/GWC_coupled/phreeqc/index.html
MINTEQA2
http://www.epa.gov/ceampubl/mmedia/minteq/index.htm
Visual MIn REQ
http://www.lwr.kth.se/english/OurSoftware/Vminteq/
MINTEQA2 for Windows
http://www.allisongeoscience.com/MINTEQ.htm
CHESS
http://chess.ensmp.fr/
Geochemists Workbench
http://www.geology.uiuc.edu/Hydrogeology/hydro_gwb.htm
GEM-Selektor
http://gems.web.psi.ch/
THERMOCALC
http://rock.esc.cam.ac.uk/astaff/holland/thermocalc.html
HCh
http://www1.geol.msu.ru/deps/geochems/soft/index_e.html
图3 HCh程序交互方案(据Shvarov, 2008; Wang et al., 2022修改) a. 氟化钪络合物和氢氧化钪络合物关于aH+和aF-的活度图解; b. 流体-岩石相互作用模型 (注:细箭头表示信息传递,双箭头表示控制传递;其他转换结果显示为虚线矩形)
Fig. 3 Interaction scheme of components of the HCh (modified after Shvarov, 2008; Wang et al., 2022) a. Activity diagrams of scandium fluoride and scandium hydroxide complexes for aH+ and aF-; b. Fluid-rock interaction model (Thin arrows correspond to information trans-fer, and double arrows show the transfer of control. Additional components are shown as dashed-line rectangles)
3.5成矿过程模拟成矿过程的本质是一系列复杂的物理化学过程。成矿过程模拟主要是基于地质观察和热力学数据,模拟成矿流体在经历降温、流体混合、水岩反应等过程时所运移的金属元素溶解度变化及矿石矿物结晶沉淀的过程。本研究主要基于HCh软件如何开展上述模拟过程进行简要介绍,并且结合钪矿床的研究实例针对水岩反应过程进行具体示范说明。
开展模拟降温成矿过程,第一步基于详细的地质观察分析,确定成矿流体成分组成、温度、压力。第二步通过HCh软件中“System”选项确定成矿流体初始元素。第三步通过“Blank”选项确定成矿流体中可能涉及的络合物。第四步通过“Input”选项确定成矿流体中所含成矿金属元素浓度。第五步通过“Control”选项确定成矿流体起始温度和降温梯度。第六步通过“Result”选项分析降温过程对成矿流体中金属溶解度的具体影响机制。模拟流体混合成矿过程前三步与降温过程相似,第四步通过“Input”选项确定成矿流体中金属元素浓度和与其混合流体的流体成分。第五步通过“Control”选项确定两者流体混合比例、温度等具体混合过程。第六步通过“Result”选项分析具体成矿机制。模拟水岩反应过程通过Input选项确定成矿流体中金属元素浓度与新鲜未蚀变围岩成分,通过“Control”选项确定成矿流体的温度、压力、氧逸度等物理化学条件后,模拟成矿流体沿着围岩中流体通道进行运移或者沉淀成矿,最终通过“Result”选项分析具体成矿机制。
针对稀土矿床中钪金属的富集成矿,Wang等(2022)基于钛合金反应釜溶解度实验确定了含氟热液流体中钪元素的溶解度和主要络合形式,并开展了成矿过程模拟。首先,需要明确含氟流体与钪含量的相关性,对此模拟250℃和500bar条件下Sc2O3(s)在1mol/kg NaCl初始流体中的溶解,在此过程F-以2×10-6逐级加入。第一步,设置流体成分为1 mol/kg NaCl,pH为3的流体与Sc2O3(s)反应,形成饱和溶液,由此可知在不含氟的条件下,初始含钪流体中的钪浓度。第二步,向体系中加入F-,随着F含量的增加含钪络合物呈现不同的变化趋势,通过上述模拟过程可获得在此条件下含氟流体迁移钪的最大含量。现结合其水岩反应成矿过程模拟实例进行示范说明。首先,成矿流体成分是通过设定1mol/kg NaCl,氟含量2000×10-6的含钪饱和流体(1.2×10-6),在250℃,500bar,pH为9.15的条件下与碳酸盐岩反应确定。其次,选用含90%白云石和10%氟磷灰石的白云岩代表碳酸盐岩。第三步,设定100 g成矿流体与1 kg碳酸盐岩围岩进行反应,并回收反应后的固体矿物。第四步,设定100 g成矿流体与第三步中反应后剩余矿物反应。第五步,重复第四步。第六步,分析上述模拟结果。Wang等(2022)通过上述模拟发现在此条件下Sc-F络合物更稳定占主导地位,研究结果表明含氟流体在250℃、500 bar和pH为3的条件下可以溶解高达2.9 ×10-6的钪,提出水岩反应是形成具有经济价值钪矿床的重要过程。同时基于该研究进一步探讨了含氯体系中Sc的迁移机制(Wang et al., 2023)。
基于钛合金反应釜溶解度实验和成矿过程模拟,有效助推了钨矿床(Wang et al., 2019)和钼矿床(Shang et al., 2020)成矿机制研究。综上所述,基于溶解度实验和成矿过程模拟研究为揭示成矿机制提供重要实验依据,对助推找矿预测工作有重要意义。
4主要认识与展望开展热液成矿模拟实验研究有助于理解认识金属成矿过程,钛合金反应釜溶解度实验是研究金属在热液流体中地球化学行为的有效手段。在充分认识该方法研究现状、原理及应用的基础上,结合当前关于钪多金属成矿的研究现状和关键金属富集机制等科学问题,笔者提出以下几点认识与展望:
(1)基于钛合金反应釜的溶解度实验方法可以满足实验溶液的高效回收,对实验过程物化条件的精准把控,以及低温下氧逸度的有效控制,将该方法与ICP-MS结合测定组分含量,计算热力学参数更全面、更精准,在热力学数据库的建立与热液成矿模型的理论研究方面有突出优势。
(2)通过溶解度实验研究获取不同物化条件下元素的络合形式和相关热力学参数,可以了解金属元素在流体中的迁移行为,进一步开展成矿模拟,为金属超常富集成矿机制研究提供关键的科学依据,有望助推战略性关键金属成矿理论研究取得突破性认识。
(3)当前,通过模拟建立理想化模型是结合地质事实简化后的单元体系,下一步需要增加多元组分的探讨,以支撑复杂热液体系中金属元素的溶解度与络合物种形的研究。基于最简化模型的有效外推区间,也需要根据地质条件进行限定以保证数据的普适性。
致 谢感谢两位匿名审稿专家就当前热液体系实验研究现状与成矿过程模拟提出的专业修改意见和建议,在此表示衷心的感谢。
-
参考文献
Benfatto M, Angelo D P, Longa D S and Pavel N V. 2002. Evidence of distorted fivefold coordination of the Cu2+ aqua ion from an X-ray-absorption spectroscopy quantitative analysis[J]. Physical Review B, 65(17):174205.
Brugger J, Etschmann B, Liu W, Testemale D, Hazemann J L, Emerich H, Van Beek W and Proux O. 2007. An XAS study of the structure and thermodynamics of Cu(I) chloride complexes in brines up to high temperature (400°C, 600bar)[J]. Geochimica et Cosmochimica Acta, 71(20): 4920-4941.
Brugger J, Liu W, Etschmann B, Mei Y, Sherman D M and Testemale D. 2016. A review of the coordination chemistry of hydrothermal systems, or do coordination changes make ore deposits[J]? Chemical Geology, 447: 219-253.
Bunker G. 2010. Introduction to XAFS: A practical guide to X-ray absorption fine structure spectroscopy[M]. New York: Cambridge University Press. 1-258.
Chen J. 2019. Metallogenesis and effective utilization of strategic-critical metals[J]. Science & Technology Review, 37(24): 1-5(in Chinese with English abstract).
Chou I M. 2021a. In situ observations and quantitative Raman spectroscopic analyses of samples in high-pressure optical cells in hydrothermal experiments[J]. Science Bulletin, 66(19): 1933-1935.
Chou I M, Wang R and Fang J. 2021b. In situ redox control and Raman spectroscopic characterization of solutions below 300℃[J]. Geochemical Perspectives Letters, 20: 1-5.
Cottis R A, Shreir L L and Burstein G T. 2010. Shreir’s corrosion: 1.01-Chemical thermodynamics[M]. Oxford: Elsevier. 1-12.
Cusanelli A, Frey U, Richens D T and Merbach A E. 1996. The slowest water exchange at a Homoleptic Mononuclear Metal Center: Variable-Temperature and variable-pressure 17O NMR study on [Ir(H2O)6]3+[J]. Journal of the American Chemical Society, 118(22): 5265-5271.
Dennis J E and Woods D J. 1987. New computing environments: Microcomputers in large-scale computing [J]. SIAM, Philadelphia, 116-122.
Dove M T. 2002. An introduction to the use of neutron scattering methods in mineral sciences[J]. European Journal of Mineralogy, 14(2): 203-224.
Duan Z H. 2010. Geological fluid state equation[J]. Scientia Sinica (Terrae), 40(4):393-413(in Chinese).
Efthimiopoulos I, Jahn S, Kuras A, Schade U and Koch-Müller M. 2017. Combined high-pressure and high-temperature vibrational studies of dolomite: Phase diagram and evidence of a new distorted modification[J]. Physics and Chemistry of Minerals, 44: 465-476.
Etschmann B E, Liu W, Mayanovic R, Mei Y, Heald S, Gordon R and Brugger J. 2019. Zinc transport in hydrothermal fluids: On the roles of pressure and sulfur vs. chlorine complexing[J]. American Mineralogist, 104(1): 158-161.
Etschmann B E, Liu W, Pring A, Grundler P V, Tooth B, Borg S, Testemale D, Brewe D and Brugger J. 2016. The role of Te(Ⅳ) and Bi(Ⅲ) chloride complexes in hydrothermal mass transfer: An X-ray absorption spectroscopic study[J]. Chemical Geology, 425: 37-51.
Etschmann B E, Liu W, Testemale D, Müller H, Rae N A, Proux O, Hazemann J L and Brugger J. 2010. An in situ XAS study of copper(Ⅰ) transport as hydrosulfide complexes in hydrothermal solutions (25-592℃, 180-600bar): Speciation and solubility in vapor and liquid phases[J]. Geochimica et Cosmochimica Acta, 74(16): 4723-4739.
Gillet P. 1996. Raman spectroscopy at high pressure and high temperature. Phase transitions and thermodynamic properties of minerals[J]. Physics and Chemistry of Minerals, 23: 263-275.
Grundler P V, Brugger J, Etschmann B E, Helm L, Liu W, Spry P G, Tian Y, Testemale D and Pring A. 2013. Speciation of aqueous tellurium(Ⅳ) in hydrothermal solutions and vapors, and the role of oxidized tellurium species in Te transport and gold deposition[J]. Geochimica et Cosmochimica Acta, 120: 298-325.
Grunwaldt J D, Wandeler R and Baiker A. 2003. Supercritical fluids in catalysis: Opportunities of in situ spectroscopic studies and monitoring phase behavior[J]. Catalysis Reviews, 45(1): 1-96.
Guan Q, Mei Y, Etschmann B, Testemale D, Louvel M and Brugger J. 2020. Yttrium complexation and hydration in chloride-rich hydrothermal fluids: A combined ab initio molecular dynamics and in situ X-ray absorption spectroscopy study[J]. Geochimica et Cosmochimica Acta, 281: 168-189.
Guo H, Xia Y, Bai R, Zhang X and Huang F. 2020. Experiments on Cu-isotope fractionation between chlorine bearing fluid and silicate magma: Implications for fluid exsolution and porphyry Cu depo-sits[J]. National Science Review, 7(8):1319-1330.
Han H and Gysi A P. 2022. UV-Vis spectrophotometric determination of the hydrolysis constants of erbium at near-neutral to alkaline pH and 25℃[C]. Goldschmidt: Hawaii, USA.
He J.1993. Some problems in the study of epithermal deposit geoche-mistry[J]. Journal of Sichuan Building Materials College, 8(1): 48-53(in Chinese with English abstract).
Helgeson H C. 1969. Thermodynamics of hydrothermal systems at elevated temperatures and pressures[J]. American Journal of Science, 267(7): 729-804.
Helgeson H C. 1981. Prediction of the thermodynamic properties of electrolytes at high pressures and temperatures[J]. Physics and Chemistry of the Earth, 13-14: 133-177.
Hu R Z, Wei W F, Bi X W, Peng J T, Qi Y Q, Wu L Y and Chen Y W. 2012. Molybdenite Re-Os and muscovite 40Ar/39Ar dating of the Xihuashan tungsten deposit, central Nanling district, South China[J]. Lithos, 150: 111-118.
Hunter E A, Hunter J R, Zajacz Z, Keith J D, Hann N L, Christiansen E H and Dorais M J. 2020. Vapor transport and deposition of Cu-Sn-Co-Ag alloys in vesicles in mafic volcanic rocks[J]. Econ. Geol., 115(2): 279-301.
Hurtig N C, Migdisov A A and Williams-Jones A E. 2021. Are vapor-like fluids viable ore fluids for Cu-Au-Mo porphyry ore formation[J] ? Econ. Geol., 116 (7): 1599-1624.
Jiang S Y and Wang W. 2022. How did supernormal enrichment of strategic key metals occur[J] ? Earth Science, 47(10): 3869-3871(in Chinese).
Lange B, Scholz F, Bautsch H J, Damaschun F and Wappler G. 1993. Thermodynamics of the xanthoconite-proustite and pyrostilpnite-pyrargyrite phase transition as determined by abrasive stripping voltammetry[J]. Physics and Chemistry of Minerals, 19: 486-491.
Liebscher A, Meixner A, Romer R L and Heinrich W. 2007. Experimental calibration of the vapour-liquid phase relations and lithium isotope fractionation in the system H2O-LiCl at 400℃ 20-28 MPa[J]. Geofluids, 7: 369-375.
Lin S, Liu Q and Li H. 2014. Electrochemical behavior of pyrite in acidic solution with different concentrations of NaCl[J]. Chinese Journal of Geochemistry, 33(4), 374-381.
Liu J F and Xu D M.1984.Preliminary experimental study of the type of migration of uranium in hydrothermal solution[J]. Journal of Mineralogy and Petrology, 70-78(in Chinese with English abstract).
Li J K, Zhang D H and Li S H. 2011. Application of melt inclusions to estimating ore-forming pressure (depth) of granite-related ore deposits[J]. Mineral Deposits, 30(6): 1002-1016(in Chinese with English abstract).
Liu W, Borg S J, Testemale D, Etschmann B, Hazemann JL and Brugger J. 2011. Speciation and thermodynamic properties for cobalt chloride complexes in hydrothermal fluids at 35-440 °C and 600 bar: An in-situ XAS study[J]. Geochimica et Cosmochimica Acta, 75(5): 1227-1248.
Liu W, Etschmann B, Foran G, Shelley M and Brugger J. 2007. Deri-ving formation constants for aqueous metal complexes from XANES spectra: Zn2+ and Fe2+ chloride complexes in hypersaline solutions[J]. American Mineralogist, 92 (5-6): 761-770.
Liu W, Etschmann B, Mei Y, Guan Q, Testemale D and Brugger J. 2020. The role of sulfur in molybdenum transport in hydrothermal fluids: Insight from in situ synchrotron XAS experiments and molecular dynamics simulations[J]. Geochimica et Cosmochimica Acta, 290: 162-179.
Liu W, Etschmann B, Migdisov A, Boukhalfa H, Testemale D, Müller H, Hazemann J L and Brugger J. 2017. Revisiting the hydrothermal geochemistry of europium(Ⅱ/Ⅲ) in light of new in-situ XAS spectroscopy results[J]. Chemical Geology, 459: 61-74.
Mao J W, Ouyang H G, Song S W, Santosh M, Yuan S D, Zhou Z H, Zheng W, Liu H, Liu P, Cheng Y B and Chen M H. 2019. Geology and metallogeny of tungsten and tin deposits in China[J]. Society of Economic Geologists Special Publication, 22: 411-482.
Mao J W, Yuan S D, Xie G Q, Song S W, Zhou Q, Gao Y B, Liu X, Fu X F, Cao J, Zeng Z L, Li T G and Fan X. 2019. New advances on metallogenic studies and exploration on critical minerals of China in 21st century[J]. Mineral Deposits, 38(5):935-969(in Chinese with English abstract).
Mao J W, Zheng W, Xie G Q, Lehmann B and Goldfarb R. 2021. Recognition of a Middle-Late Jurassic arc-related porphyry copper belt along the southeast China coast: Geological characteristics and metallogenic implications[J]. Geology, 49(5): 592-596.
Mei Y, Liu W, Sherman D M and Brugger J. 2014. Metal complexation and ion hydration in low density hydrothermal fluids: Ab initio molecular dynamics simulation of Cu(Ⅰ) and Au(Ⅰ) in chloride solutions (25~1000°C, 1~5000bar)[J]. Geochimica et Cosmochimica Acta, 131: 196-212.
Mei Y, Etschmann B, Liu W, Sherman D M, Testemale D and Brugger J. 2016. Speciation and thermodynamic properties of zinc in sulfur-rich hydrothermal fluids: Insights from ab initio molecular dynamics simulations and X-ray absorption spectroscopy[J]. Geochimica et Cosmochimica Acta, 179: 32-52.
Migdisov A A and Williams-Jones A E. 2007. An experimental study of the solubility and speciation of neodymium (Ⅲ) fluoride in F-bearing aqueous solutions[J]. Geochimica et Cosmochimica Acta, 71(12): 3056-3069.
Migdisov A A, Bychkov A Y, Williams-Jones A E and Van Hinsberg V J. 2014a. A predictive model for the transport of copper by HCl-bearing water vapour in ore-forming magmatic-hydrothermal systems: Implications for copper porphyry ore formation[J]. Geochimica et Cosmochimica Acta, 129: 33-53.
Migdisov A A and Williams-Jones A E. 2014b. Hydrothermal transport and deposition of the rare earth elements by fluorine-bearing aqueous liquids[J]. Mineralium Deposita, 49: 987-997.
Migdisov A A, Williams-Jones A E, Brugger J and Caporuscio F A. 2016. Hydrothermal transport, deposition, and fractionation of the REE: Experimental data and thermodynamic calculations[J]. Chemical Geology, 439: 13-42.
Neilson G W, Mason P E, Ramos S and Sullivan D. 2001. Neutron and X-ray scattering studies of hydration in aqueous solutions[J]. Philosophical Transactions of the Royal Society A: Mathematical, Physical and Engineering Sciences, 359(1785): 1575-1591.
Nelder J A and Mead R. 1965. A simplex method for function minimization[J]. The Computer Journal, 7(4): 308-313.
Nisbet H, Migdisov A A, Xu H, Guo X, Hinsberg V V, Williams-Jones A E, Boukhalfa H and Roback R. 2018. An experimental study of the solubility and speciation of thorium in chloride-bearing aqueous solutions at temperatures up to 250℃[J]. Geochimica et Cosmochimica Acta, 239: 363-373.
Oelkers E H, Bénézeth P and Pokrovski G S. 2009. Thermodynamic Databases for Water-Rock Interaction[J]. Reviews in Mineralogy and Geochemistry, 70: 1-46.
Pokrovski G S and Dubessy J. 2015. Stability and abundance of the trisulfur radical ion S3−in hydrothermal fluids[J]. Earth and Planetary Science Letters, 411: 298-309.
Pokrovski G S, Martin F, Hazemann J L and Schott J. 2000. An X-ray absorption fine structure spectroscopy study of germanium-organic ligand complexes in aqueous solution[J]. Chemical Geology, 163(1-4): 151-165.
Pokrovski G S, Roux J and Harrichoury J C. 2005a. Fluid density control on vapor-liquid partitioning of metals in hydrothermal systems[J]. Geology, 33(8): 657-660.
Pokrovski G S, Roux J, Hazemann J L and Testemale D. 2005b. An X-ray absorption spectroscopy study of argutite solubility and aqueous Ge(Ⅳ) speciation in hydrothermal fluids to 500℃ and 400 bar[J]. Chemical Geology, 217(1-2): 127-145.
Pokrovski G S, Borisova A Y, Roux J, Hazemann J L, Petdang A, Tella M and Testemale D. 2006. Antimony speciation in saline hydrothermal fluids: A combined X-ray absorption fine structure spectroscopy and solubility study[J]. Geochimica et Cosmochimica Acta, 70(16): 4196-4214.
Pokrovski G S, Tagirov B R, Schott J, Hazemann J L and Proux O. 2009. A new view on gold speciation in sulfur-bearing hydrothermal fluids from in situ X-ray absorption spectroscopy and quantum-chemical modeling[J]. Geochimica et Cosmochimica Acta, 73(18): 5406-5427.
Pokrovski G S, Borisova A Y and Harrichoury J C. 2008. The effect of sulfur on vapor-liquid fractionation of metals in hydrothermal systems[J]. Earth and Planetary Science Letters, 266(3-4): 345-362.
Pokrovski G S, Roux J, Ferlat G, Jonchiere R, Seitsonen A P, Vuilleumier R and Hazemann J L. 2013. Silver in geological fluids from in situ X-ray absorption spectroscopy and first-principles molecular dynamics[J]. Geochimica et Cosmochimica Acta, 106: 501-523.
Qi D, Behrens, H, Botcharnikov R, Derrey I, Holtz F, Zhang C, Li X and Horn I. 2020. Reaction between Cu-bearing minerals and hydrothermal fluids at 800℃ and 200 MPa: Constraints from synthetic fluid inclusions[J]. American Mineralogist, 105(8): 1126-1139.
Reynard B and Guyot F. 1994. High-temperature properties of geikielite (MgTiO3-ilmenite) from high-temperature high-pressure Raman spectroscopy: Some implications for MgSiO3-ilmenite[J]. Physics and Chemistry of Minerals, 21: 441-450.
Rinaldi R, Gatta G D, Artioli G, Knight K S and Geiger C A. 2005. Crystal chemistry, cation ordering and thermoelastic behaviour of CoMgSiO4 olivine at high temperature as studied by in situ neutron powder diffraction[J]. Physics and Chemistry of Minerals, 32: 655-664.
Rinaldi R. 2002. Neutron scattering in mineral sciences[J]. European Journal of Mineralogy, 14(2): 195-202.
Schmidt C, Gottschalk M, Zhang R, and Lu J. 2021. Oxygen fugacity during tin ore deposition from primary fluid inclusions in cassiterite[J]. Ore Geology Reviews, 139, 104451.
Scholten L, Schmidt C, Lecumberri-Sanchez P, Newville M, Lanzirotti A, Sirbescu M L C and Steele-MacInnis M. 2019. Solubility and speciation of iron in hydrothermal fluids[J]. Geochimica et Cosmochimica Acta, 252: 126-143.
Shang L B, Williams-Jones A E, Wang X S, Timofeev A, Hu R Z and Bi X W. 2020. An experimental study of the solubility and speciation of MoO3(s) in hydrothermal fluids at temperatures up to 350℃[J]. Econ. Geol., 115 (3): 661-669.
Shvarov Y V. 2008. HCh: New potentialities for the thermodynamic simulation of geochemical systems offered by Windows[J]. Geochemistry International, 46(8): 834.
Suleimenov O and Seward T. 2000. Spectrophotometric measurements of metal complex formation at high temperatures: The stability of Mn(Ⅱ) chloride species[J]. Chemical Geology, 167(1-2): 177-192.
Testemale D, Hazemann J L, Pokrovski G S, Joly Y, Roux J, Argoud R and Geaymond O. 2004. Structural and electronic evolution of the As(OH)3 molecule in high temperature aqueous solutions: An X-ray absorption investigation[J]. The Journal of Chemical Physics, 121(18): 8973-8982.
Timofeev A, Migdisov A A and Williams-Jones A E. 2015. An experimental study of the solubility and speciation of niobium in fluoride-bearing aqueous solutions at elevated temperature[J]. Geochimica et Cosmochimica Acta, 158: 103-111.
Timofeev A, Migdisov A A and Williams-Jones A E. 2017. An experimental study of the solubility and speciation of tantalum in fluoride-bearing aqueous solutions at elevated temperature[J]. Geochimica et Cosmochimica Acta, 197: 294-304.
Timofeev A, Migdisov A A and Williams-Jones A E. 2018. Uranium transport in acidic brines under reducing conditions[J]. Nature Communications, 9: 1469.
Voigt A, Cassidy M, Castro J M, Pyle D M, Mather T A, Helo C, Abdurrachman M and Kurniawan IA. 2022. Experimental investigation of trachydacite magma storage prior to the 1257 eruption of Mt Samalas[J]. Journal of Petrology, 63(8): egac066.
Wang J L, Wei H Z, Williams-Jones A E, Dong G, Zhu Y F, Jiang S Y, Ma J, Hohl S V, Liu X, Li Y C and Lu J J. 2022. Silver isotope fractionation in ore-forming hydrothermal systems[J]. Geochimica et Cosmochimica Acta, 322: 24-42.
Wang J X, Williams-Jones A E, Timofeev A, Liu J J and Yuan S D. 2022. An experimental investigation of the solubility and speciation of scandium in fluoride-bearing aqueous liquids at temperatures up to 250℃[J]. Geochimica et Cosmochimica Acta, 330: 67-79.
Wang J X, Williams-Jones A E, Timofeev A, Zhang X N, Liu J J, Yuan S D. 2023. The role of scandium chloride and hydroxide complexes in the formation of scandium deposits: Insights from experiments and modeling[J]. Econ. Geol., 118 (8): 1995-2004.
Wang X S, Timofeev A, Williams-Jones A E, Shang L B and Bi X W. 2019. An experimental study of the solubility and speciation of tungsten in NaCl-bearing aqueous solutions at 250, 300, and 350℃[J]. Geochimica et Cosmochimica Acta , 265: 313-329.
Wang X S, Williams-Jones A E, Hu R Z, Shang L B and Bi X W. 2021. The role of fluorine in granite-related hydrothermal tungsten ore genesis: Results of experiments and modeling[J]. Geochimica et Cosmochimica Acta, 292: 170-187.
Waychunas G A and Brown G E. 1990. Polarized X-ray absorption spectroscopy of metal ions in minerals[J]. Physics and Chemistry of Minerals, 17: 420-430.
Wen H J, Zhou Z B, Zhu C W, Luo C G, Wang D Z, Du S J, Li X F, Chen M H and Li H Y. 2019. Critical scientific issues of super-enrichment of dispersed metals[J]. Acta Petrologica Sinica, 35(11):3271-3291(in Chinese with English abstract).
Wen H J, Zhu C W, Du S J, Fan Y and Luo C G. 2020. Gallium(Ga), germanium(Ge), thallium(Tl) and cadmium(Cd) resources in China[J]. Chinese Science Bulletin, 65(33):3688-3699(in Chinese with English abstract).
White W M. 2013. Geochemistry[M]. Chichester: John Wiley & Sons. 1-660.
Williams-Jones A E and Migdiso A A. 2014. Experimental constraints on the transport and deposition of metals in ore-forming hydrothermal systems[M]. Building Exploration Capability for the 21st Century, Kelley K D and Golden H C. U S: Society of Economic Geologists. 18: 77-95.
Williams-Jones A E, Migdisov A A and Samson I M. 2012. Hydrothermal mobilisation of the rare earth elements: A Tale of "Ceria" and "Yttria"[J]. Elements, 8(5): 355-360.
Xia Y H, Wu R,Zhang Y F,Liu S Q, Du H L, Han J Z, Wang C S, Chen X P, Xie L, Yang Y C and Yang J B. 2017. Tunable giant exchange bias in the single-phase rare-earth-transition-metal intermetallics YMn12-xFexwith highly homogenous intersublattice exchange coupling[J]. Physical Review B, 96(6): 064440.
Xiong X L, Hou T and Wang X L. 2022. Advances and perspectives of experimental metallogeny[J]. Earth Science, 47(8): 2701-2713(in Chinese with English abstract).
Yin Y and Zajacz Z. 2018. The solubility of silver in magmatic fluids: Implications for silver transfer to the magmatic-hydrothermal ore-forming environment[J]. Geochimica et Cosmochimica Acta, 238: 235-251.
Yuan S D, Chou I M, Burruss R C, Wang X L and Li J K. 2013. Disoportionation and thermochemical sulfate reduction reactions in S-H2O-CH4 and S-D2O-CH4 systems from 200 to 340℃ at elevated pressures[J]. Geochimica et Cosmochimica Acta, 118: 263-275.
Yuan S D, Ellis G S, Chou I M and Burruss R C. 2017. Experimental investigation on thermochemical sulfate reduction in the presence of 1-pentanethiol at 200 and 250℃: Implications for in situ TSR processes occurring in some MVT deposits[J]. Ore Geology Reviews, 91:57-65.
Yuan S D, Williams-Jones A E, Mao J W, Zhao P L, Yan C and Zhang D L. 2018. The origin of the Zhangjialong tungsten deposit, South China: Implications for W-Sn mineralization in large granite batholiths[J]. Econ. Geol., 113(5): 1193-1208.
Yuan S D, Williams-Jones A E, Romer R L, Zhao P L and Mao J W. 2019. Protolith-related thermal controls on the decoupling of Sn and W in Sn-W metallogenic provinces: Insights from the Nanling region, China[J]. Econ. Geol., 114(5): 1005-1012.
Yuan S D, Zhao P L and Liu M. 2020. Some problems involving in petrogenesis and metallogenesis of granite-related tin deposits[J]. Mineral Deposits, 39(4): 607-618(in Chinese with English abstract).
Yuan S D and Zhao P L. 2021. New synthetic fluid inclusion method to investigate partition behavior of ore metals between melt and fluid phases[J]. Scientia Sinica Terrae, 51(2): 241-249(in Chinese with English abstract).
Zajacz Z, Halter W E, Pettke T and Guillong M. 2008. Determination of fluid/melt partition coefficients by LA-ICPMS analysis of co-existing fluid and silicate melt inclusions: Controls on element partitioning[J]. Geochimica et Cosmochimica Acta, 72(8): 2169-2197.
Zajacz Z, Seo J H, Candela P A, Piccoli P M, Heinrich C A and Guillong M. 2010. Alkali metals control the release of gold from volatile-rich magmas[J]. Earth and Planetary Science Letters, 297(1-2): 50-56.
Zajacz Z, Candela P A, Piccoli P M, Sanchez-Valle C and Wälle M. 2013. Solubility and partitioning behavior of Au, Cu, Ag and reduced S in magmas[J]. Geochimica et Cosmochimica Acta, 112: 288-304.
Zajacz Z, 2015. The effect of melt composition on the partitioning of oxidized sulfur between silicate melts and magmatic volatiles[J]. Geochimica et Cosmochimica Acta, 158: 223-244.
Zhai Y S. 1999 .On the metallogenic system[J]. Earth Science Frontiers, 6(1):14-28(in Chinese with English abstract).
Zhang S and Liu Y S. 1995. Experimental study of gold solubility and its geological significance[J]. Geochimica, (S1):168-176(in Chinese with English abstract).
Zhao P L, Zajacz Z, Tsay A and Yuan S D. 2022a. Magmatic-hydrothermal tin deposits form in response to efficient tin extraction upon magma degassing[J]. Geochimica et Cosmochimica Acta, 316(1): 331-346.
Zhao P L, Zajacz Z, Tsay A, Chu X, Cheng Q M and Yuan S D. 2022b. The partitioning behavior of Mo during magmatic fluid exsolution and its implications for Mo mineralization[J]. Geochimica et Cosmochimica Acta, 339: 115-126.
Zhao P L, Chu X, Williams-Jones A E, Mao J W and Yuan S D. 2022c. The role of phyllosilicate partial melting in segregating W and Sn deposits in W-Sn metallogenic provinces[J]. Geology, 50(1): 121-125.
Zhao P L, Yuan S D, Williams-Jones A E, Romer R L, Yan C, Song S W and Mao J W. 2022d. Temporal separation of W and Sn mine-ralization by temperature-controlled incongruent melting of a single protolith: Evidence from the Wangxianling area, Nanling region, South China[J]. Econ. Geol., 117(3): 667-682.
Zhao X L, Li H P and Su G L. 2013. A progress on quartz solubility in aqueous fluids at high temperature and high pressure[J]. Acta Mineralogica Sinica, 33(1):75-82(in Chinese with English abstract)
Zhong R, Brugger J, Chen Y and Li W. 2015. Contrasting regimes of Cu, Zn and Pb transport in ore-forming hydrothermal fluids[J]. Chemical Geology, 395, 154-164.
Zhou L, Mavrogenes J, Spandler C and Li H. 2016. A synthetic fluid inclusion study of the solubility of monazite-(La) and xenotime-(Y) in H2O-Na-K-Cl-F-CO2 fluids at 800°C and 0.5 GPa[J]. Chemical Geology, 442: 121-129.
附中文参考文献
陈骏. 2019.关键金属超常富集成矿和高效利用[J].科技导报, 37(24):1-5.
段振豪. 2010.地质流体状态方程[J].中国科学:地球科学, 40(4):393-413.
何江. 1993.低温矿床成矿地球化学研究的某些问题[J].四川建材学院学报, 8(1): 48-53.
蒋少涌,王微. 2022.战略性关键金属是如何发生超常富集成矿的[J].地球科学, 47(10):3869-3871.
李建康,张德会,李胜虎. 2011.熔体包裹体在估算花岗岩类矿床形成压力(深度)方面的应用[J] .矿床地质, 30(6):1002-1016.
刘吉芳,徐德明. 1984.热液中铀迁移形式的初步实验研究[J].矿物岩石, (1): 70-78.
毛景文,袁顺达,谢桂青,宋世伟,周琦,高永宝,刘翔,付小方,曹晶,曾载淋,李通国,樊锡银. 2019. 21世纪以来中国关键金属矿产找矿勘查与研究新进展[J].矿床地质, 38(5):935-969.
温汉捷,周正兵,朱传威,罗重光,王大钊,杜胜江,李晓峰,陈懋弘,李红谊. 2019.稀散金属超常富集的主要科学问题[J].岩石学报, 35(11):3271-3291.
温汉捷,朱传威,杜胜江,范裕,罗重光. 2020.中国镓锗铊镉资源[J].科学通报, 65(33):3688-3699.
熊小林,侯通,王小林. 2022.实验矿床学的发展现状和前景展望[J].地球科学, 47(8): 2701-2713.
袁顺达,赵盼捞,刘敏. 2020.与花岗岩有关锡矿成岩成矿作用研究若干问题讨论[J].矿床地质, 39(4):607-618.
袁顺达,赵盼捞. 2021.基于新的合成流体包裹体方法对成矿金属在熔体-流体相间分配行为的实验研究[J].中国科学:地球科学, 51(2):241-249.
翟裕生. 1999.论成矿系统[J].地学前缘, 6(1):14-28.
张生,刘玉山. 1995.金溶解度实验研究及地质意义[J].地球化学, (S1): 168-176.
赵晓莉,李和平,苏根利. 2013.高温高压下水流体中石英溶解度研究进展[J].矿物学报, 33(1):75-82.
摘要
热液流体对金属元素的迁移、富集及沉淀成矿起着关键作用,定量刻画金属元素在热液成矿过程中的运移是矿床学研究的重点和难点。以热力学理论为基础,通过开展溶解度实验,揭示金属元素在热液流体中的地球化学行为,是研究金属元素迁移、富集沉淀机制行之有效的重要手段。其中,基于钛合金反应釜开展不同物理化学条件下金属元素在热液流体中的溶解度实验,结合相关的热力学模拟,能够精确测定金属元素在热液流体中的溶解度、络合物配位数、气液两相中的分配系数及相关热力学参数,为定量模拟热液成矿过程提供重要的实验依据。文章归纳了近年来针对热液体系的实验矿床学方法,分析了各类实验方法与分析手段的优点与不足。重点介绍钛合金反应釜溶解度实验和热力学模拟计算方法,并结合该方法模拟钪矿床成矿的水岩反应过程,说明该方法在成矿过程模拟中的应用,最后阐述了其存在的问题,并对该领域未来的研究趋势提出了展望,以期推动该方法在战略性关键金属超常富集机制研究中的应用。
Abstract
Hydrothermal fluids play a key role in transport, enrichment and deposition of the metallic elements, and therefore, quantitatively discretion of the hydrothermal mobility of metal elements has always attracted considerable attentions. Hydrothermal experiments are one of the most effective ways to understand the behavior of the metallic elements in hydrothermal systems. The Ti-alloy-reactor solubility experiment, conducted under various physical and chemical conditions, combined with the related thermodynamic calculation, can accurately determine the solubility, the complex coordination number, the partitioning coefficient between gas-liquid phases and the other related thermodynamic parameters of metal elements. The application of these thermodynamic dataset could effectively evaluate the mechanisms responsible for hydrothermal transport and deposition of the metal elements. In this contribution, we review various hydrothermal experimental methods, and analyzes the advantages and disadvantages of various experimental and analytical methods. Combined with the study cases in scandium mineralization process, the solubility experiments conducted in titanium alloy fusion reactors are particularly introduced. This paper also summarizes current application status of this method in simulation of mineralization process, and the research trends are put forward in the end. This paper provides a comprehensive review of the Ti-alloy-reactor solubility experiments and the related thermodynamic calculation, and the existing problems of this method are explained, in order to promote the application and development of this method in simulation of the mineralization process, and so as to make a major breakthrough in the study of the mechanism of super-enrichment strategic key metals.